Lentic vs. Lotic
|
A lotic body of water is a moving body of water, such as a stream or river. A lentic body of water is a stagnant body of water, such as a pond or lake. Because decomposition is constantly taking place at the bottom of lentic systems and the constant motion of water allows oxygen to mix more easily with the water in lotic systems, lotic systems tend to have more oxygen than lentic systems.
|
Eutrophic vs. Oligotrophic
Limnology
 http://www.upstatefreshwater.org/NRT-Data/Data-Analysis/data-analysis.html
http://www.upstatefreshwater.org/NRT-Data/Data-Analysis/data-analysis.html
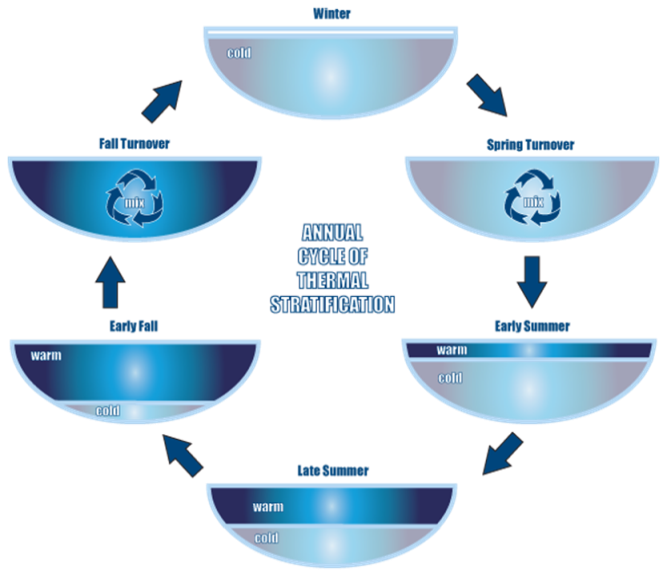
Bodies of water are composed of three layers: the epilimnion, metalimnion, and hypolimnion. The epilimnion is the topmost and warmest layer of the three. Below it, in the middle, is the metalimnion, where temperature starts to dramatically decline by 1 degree C for every meter of depth; this dramatic change in temperature is known as the thermocline. The hypolimnion, the bottom layer, is the coldest of the three layers.
Overturn
|
Lentic
As fall approaches, the weather becomes cooler to the epilimnion does as well. The cooling of the epilimnion decreases the difference in density between the epilimnion and the other layers, so they mix together, distributing nutrients and oxygen throughout the lake and making it uniform in temperature. In the winter, the lake is covered by a sheet of ice. This layer of ice cuts off from its supply of oxygen in the atmosphere, and what little oxygen remains is used up by bacteria as they decompose organic matter at the bottom of the lake. In the spring, the ice sheet melts and the lake mixes again, redistributing oxygen and nutrients throughout the lake. In the summer, the lake becomes stratified again. Lotic Lotic systems are stratified like lentic systems, but unlike lentic systems, lotic systems do not undergo seasonal overturn. Instead, because the water in lotic systems is constantly churning, lotic systems are constantly going through overturn. |
Water Quality
pH
The ideal range of pH for most freshwater fish is between 6 and 9.
Work Cited: http://www.fondriest.com/environmental-measurements/parameters/water-quality/ph/
Nitrates/Nitrites
Nitrate/Nitrite levels in bodies of water should be fairly low because excess amounts of them could poison plants and animals.
Work Cited: http://water.epa.go/type/rsl/monitoring/vms57.cfm
Dissolved Oxygen
Aquatic life is most affected by low DO levels, which increase mortality rates in aquatic organisms. Different species are more or less tolerant of low DO levels, but 7mg/L seems to be an optimal level of DO for most aquatic organisms.
Work Cited: http://www.fondriest.com/environmental-measurements/parameters/water-quality/dissolved-oxygen/
Chlorine
In freshwater, ideally there should be low dissolved Cl levels, as an excess of it and dissolved Na levels would form salt, making the water brackish, which is unsuitable for most freshwater fish.
Work Cited: http://www.ruf.rice.edu/~cbensa/Salinity/
The ideal range of pH for most freshwater fish is between 6 and 9.
Work Cited: http://www.fondriest.com/environmental-measurements/parameters/water-quality/ph/
Nitrates/Nitrites
Nitrate/Nitrite levels in bodies of water should be fairly low because excess amounts of them could poison plants and animals.
Work Cited: http://water.epa.go/type/rsl/monitoring/vms57.cfm
Dissolved Oxygen
Aquatic life is most affected by low DO levels, which increase mortality rates in aquatic organisms. Different species are more or less tolerant of low DO levels, but 7mg/L seems to be an optimal level of DO for most aquatic organisms.
Work Cited: http://www.fondriest.com/environmental-measurements/parameters/water-quality/dissolved-oxygen/
Chlorine
In freshwater, ideally there should be low dissolved Cl levels, as an excess of it and dissolved Na levels would form salt, making the water brackish, which is unsuitable for most freshwater fish.
Work Cited: http://www.ruf.rice.edu/~cbensa/Salinity/